In pharmaceutical quality systems, stability deviations are often treated as red flags pointing directly at the product. An unexpected assay shift, a dissolution drift, or an environmental excursion inside a stability chamber can quickly trigger concern that the formulation itself is unstable. In many investigations, the product becomes the prime suspect almost immediately.
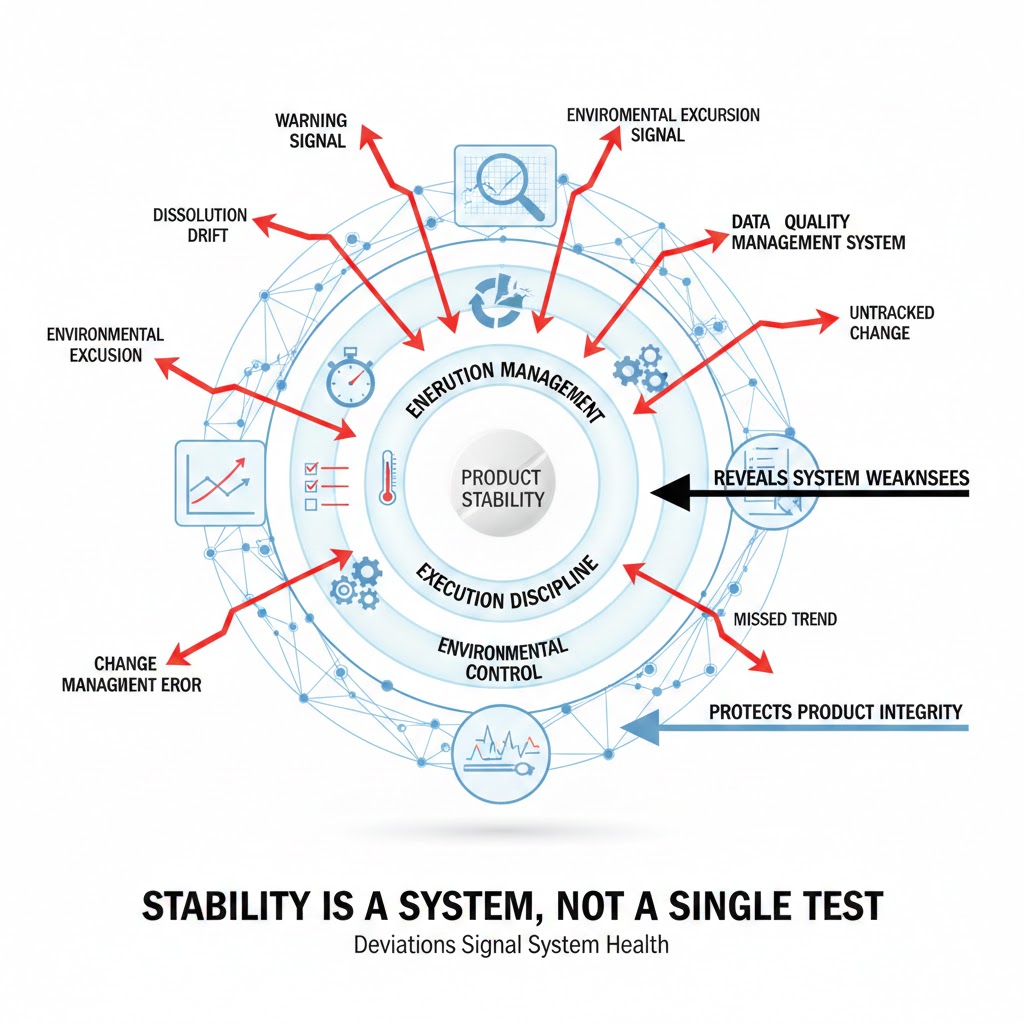
Yet, in practice, stability deviations frequently reveal weaknesses not in the dosage form—but in the systems designed to protect it.
Understanding this distinction is critical. When deviations are misattributed to the product, organizations risk overlooking deeper structural gaps in environmental control, monitoring strategies, procedural discipline, or change management. Over time, these overlooked system weaknesses become far more dangerous than the original deviation itself.
Stability Is a System, not a Single Test
A stability study does not exist in isolation. It is the outcome of multiple interconnected systems working together over months or years. The chamber must maintain consistent environmental conditions. Sensors must accurately reflect those conditions. Sampling must be controlled. Analytical methods must perform reliably. Documentation must capture events truthfully and completely. Change control must anticipate downstream impacts.
When stability data deviates from expectation, the question is not only whether the product degraded—but whether every part of this interconnected system functioned as intended.
A deviation, therefore, should be viewed as a signal from the system, not automatically as proof of product weakness.
Environmental Control: The Silent Variable
Stability chambers are engineered to create controlled temperature and humidity conditions, but they are dynamic mechanical systems. Airflow patterns evolve. Components age. Filters accumulate particulate matter. Even minor maintenance interventions can subtly alter environmental distribution.
A chamber may continue displaying “within limit” readings at its control sensor while localized variations develop elsewhere. If monitoring is limited to a small number of probes, these micro-variations may go undetected. Over time, such environmental inconsistencies can influence degradation kinetics without triggering alarms.
In these cases, the product is responding predictably to its environment. The instability is not inherent to the formulation; it arises from environmental variability that was insufficiently characterized or monitored.
The Role of Execution Discipline
Stability programs rely heavily on procedural consistency. Small variations in execution can introduce meaningful differences in data.
Consider sample handling. If samples remain outside the chamber longer than intended during pull points, they may experience transient exposure to uncontrolled conditions. If chamber doors are opened frequently or for extended periods, internal recovery times may increase. If pull points are delayed, degradation intervals shift slightly.
Each of these factors may appear minor. Individually, they may not seem capable of driving a deviation. Collectively, however, they introduce variability that can be mistaken for product instability.
The deviation, in such scenarios, reflects inconsistency in execution rather than formulation failure.
Change Management
Post-approval changes represent another common source of stability deviations. A new raw material supplier may introduce subtle variability in moisture content or impurity profile. Equipment modifications during scale-up may alter blend uniformity or compression characteristics. A packaging component may change in material density or sealing performance.
These changes often pass initial risk assessments because they do not immediately impact release specifications. However, stability is time-dependent. The true impact of a seemingly minor change may only become visible months later during long-term studies.
When stability deviations occur under these circumstances, the product has not suddenly become unstable. Instead, earlier system-level risk assessments may have underestimated long-term consequences.
Data Interpretation
Stability science is not limited to pass-or-fail outcomes. Trends carry meaning long before specifications are breached. A gradual downward assay trend, a consistent increase in degradation product formation, or subtle dissolution variability may indicate emerging environmental or process-related issues.
If data review processes focus solely on specification limits without robust trend analysis, early warning signals are missed. When a deviation eventually occurs, it may appear abrupt—but the data often tells a longer story.
In such cases, the deviation reveals a weakness in data evaluation practices rather than a sudden breakdown of product stability.
Conclusion
Pharmaceutical products exist within controlled environments shaped by engineering, procedures, and human oversight. When stability deviations occur, they often reflect the condition of this surrounding system rather than intrinsic formulation weakness.
Recognizing this distinction protects organizations from reactive decisions and encourages deeper, more meaningful investigation. Stability science is not only about chemistry—it is about systems thinking.
Ultimately, product quality is sustained not just by formulation design, but by the integrity of the systems that support it. When stability deviations appear, they should prompt a broader question: not simply “What failed in the product?” but “What does this reveal about our system?”
