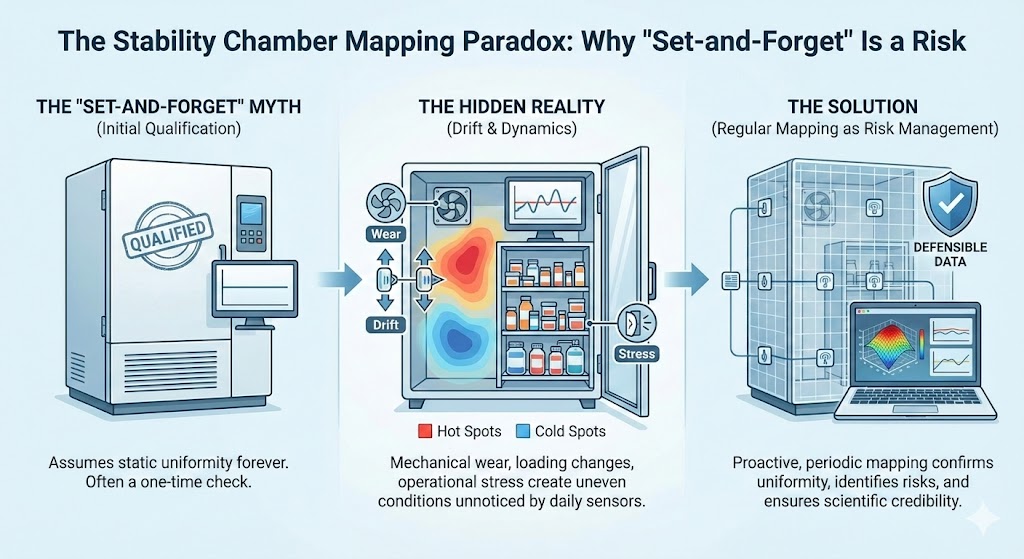
In pharmaceutical and biotech laboratories, stability chambers are often treated as “set-and-forget” systems. Once qualified, they are expected to maintain uniform temperature and humidity for years with minimal intervention. In reality, this assumption is one of the most common sources of stability risk.
Temperature and humidity mapping is not a one-time regulatory formality. It is a continuous risk-management activity that ensures the data generated inside a stability chamber remains scientifically valid and regulatorily defensible.
This article explains why stability chambers require more frequent mapping than most labs anticipate, and what is truly at stake when mapping is delayed or overlooked.
What Mapping Really Confirms (Beyond Compliance)
Stability chamber mapping is not merely about demonstrating that a chamber reaches a target setpoint such as 40°C / 75% RH or 25°C / 60% RH. Mapping confirms:
- Temperature and humidity uniformity across the usable volume
- Identification of hot spots and cold spots
- Stability of conditions over time and during operational stress
- Suitability of the chamber for long-term product exposure
Without regular mapping, there is no objective evidence that samples stored at different locations inside the chamber are experiencing equivalent conditions.
Why Initial Qualification Is Not Enough
Most chambers are mapped during installation qualification (IQ/OQ/PQ). However, several variables begin to change immediately after routine use starts.
1. Mechanical Wear and Sensor Drift
Over time, chamber components such as fans, heaters, humidifiers, and dehumidifiers degrade. Similarly, control sensors may drift between calibrations. These changes are gradual and often not visible in daily monitoring trends, yet they can significantly alter airflow and micro-climates inside the chamber.
2. Changes in Loading Patterns
Stability chambers are rarely used in the same way they were during qualification. As programs expand, chambers become more densely loaded, shelves are rearranged, and sample containers vary in size and thermal mass. Each of these changes affects airflow and heat distribution.
A chamber mapped under light or empty conditions may behave very differently when fully loaded.
3. Door Openings and Operational Stress
Routine activities such as sample pulls, inspections, and audits introduce temperature and humidity disturbances. Over time, these repeated stresses can reveal recovery issues that were not evident during initial qualification.
Mapping under dynamic conditions is the only way to verify that the chamber recovers uniformly after disturbances.
Regulatory Expectations Are Risk-Based, Not Static
Regulators do not prescribe a fixed mapping frequency for all chambers. Instead, expectations are risk-based, and inspectors typically assess:
- Chamber criticality (commercial vs development studies)
- Duration of stability studies stored
- Historical deviations or excursions
- Change history (repairs, relocations, upgrades)
- Monitoring trend behavior
In practice, this means that waiting several years between mapping exercises may be difficult to justify, especially for chambers supporting long-term or registration stability data.
Common Scenarios That Demand Re-Mapping
Many laboratories underestimate how often mapping should be repeated. Re-mapping is generally expected after:
- Chamber relocation or major repair
- Replacement of control systems or airflow components
- Significant changes in shelving or loading configuration
- Repeated excursions or unexplained alarms
- Long periods of continuous operation without downtime
Even in the absence of visible failures, periodic mapping serves as preventive verification, not reactive troubleshooting.
The Data Integrity Risk of Infrequent Mapping
Stability data assumes that all samples are exposed to defined conditions within allowable tolerances. If chamber uniformity is not periodically reconfirmed, the following risks emerge:
- Stability results may no longer represent labeled storage conditions
- OOS or OOT results become difficult to scientifically defend
- Root cause investigations lack foundational environmental evidence
- Regulatory confidence in the stability program may be compromised
In severe cases, questions around chamber performance can impact entire stability studies, not just individual time points.
Conclusion
Stability chambers do not fail suddenly; they drift slowly. Mapping is the only reliable way to detect those changes before they affect product data.
Viewing mapping as a one-time qualification activity is a technical misconception. In reality, stability chambers need mapping more often than most laboratories expect, not because regulations demand it, but because science and risk management do.
Regular mapping is not about doing more work. It is about ensuring that stability data remains credible, defensible, and scientifically sound throughout the life of a product.
