Cold chain management has become one of the most critical aspects of pharmaceutical distribution, particularly with the growth of biologics, vaccines, cell and gene therapies, and temperature-sensitive injectables. Products labeled for storage at 2–8°C or below are not simply kept cold as a precaution; their safety, potency, and stability depend directly on maintaining that temperature range.
Despite well-designed logistics systems, temperature excursions still occur. A shipment may be delayed at customs. A refrigeration unit may malfunction during transport. A package may remain on a loading dock longer than expected. When this happens, the immediate concern is often binary: Is the product still usable, or must it be discarded?
In reality, cold chain excursions are rarely that simple. What happens during an excursion depends on multiple scientific and operational factors, and effective management requires structured evaluation rather than assumption.
What Actually Happens During a Cold Chain Excursion
When a temperature-sensitive product is exposed to conditions outside its labeled storage range, the impact depends on the magnitude, duration, and direction of the deviation.
If temperatures rise above the recommended range, the most common concern is accelerated degradation. Chemical reactions generally proceed faster at higher temperatures. For small-molecule drugs, this may increase impurity formation or reduce assay over time. For biologics, elevated temperatures can lead to protein denaturation, aggregation, or loss of biological activity. However, degradation is not instantaneous. It follows kinetics influenced by formulation composition, buffer systems, excipients, and prior exposure history.
Freezing presents a different type of risk. While some products tolerate short periods at sub-zero temperatures, others are highly sensitive to freeze–thaw cycles. Ice crystal formation can disrupt protein structures, destabilize emulsions, crack prefilled syringes, or cause phase separation in suspensions. Importantly, damage from freezing may not always be visible. A vial may appear normal while subtle structural changes compromise long-term stability.
Temperature excursions also interact with cumulative stress. A product that has already spent months in distribution or near the end of its shelf life may respond differently to an excursion than a freshly manufactured batch. Stability is not only about the current exposure, but about total exposure history.
Understanding these dynamics is essential before making decisions about product disposition.
Why Not Every Excursion Results in Product Failure
A common misconception is that any deviation outside labeled storage conditions automatically renders a product unusable. In practice, labeled storage ranges are designed to ensure quality across the entire shelf life under worst-case conditions. Short, controlled excursions do not necessarily equate to immediate instability.
Many pharmaceutical products undergo stability studies at accelerated conditions precisely to understand how temperature influences degradation rates. This data can support a scientific assessment of limited excursions. The key is not whether the excursion occurred, but whether its time–temperature profile exceeds what the product has been demonstrated to tolerate.
However, making this determination requires accurate data and structured evaluation. Assumptions without documentation create regulatory risk and patient safety concerns.
The Operational Reality of Excursions
In real-world distribution networks, cold chain excursions typically stem from process breakdown rather than intentional neglect. Logistics systems involve packaging performance, route planning, environmental monitoring devices, third-party carriers, and handoffs between facilities. A weakness at any point can introduce temperature risk.
Sometimes the problem lies not in the actual exposure, but in insufficient monitoring resolution. Data loggers may record temperatures at intervals that fail to capture short spikes. Devices may be improperly positioned inside packaging, leading to readings that do not accurately represent product temperature. In other cases, alarms may not be reviewed promptly, delaying response.
The challenge is that cold chain management is not purely technical; it is procedural and human as well. Training, documentation discipline, and clear escalation pathways are just as important as packaging validation.
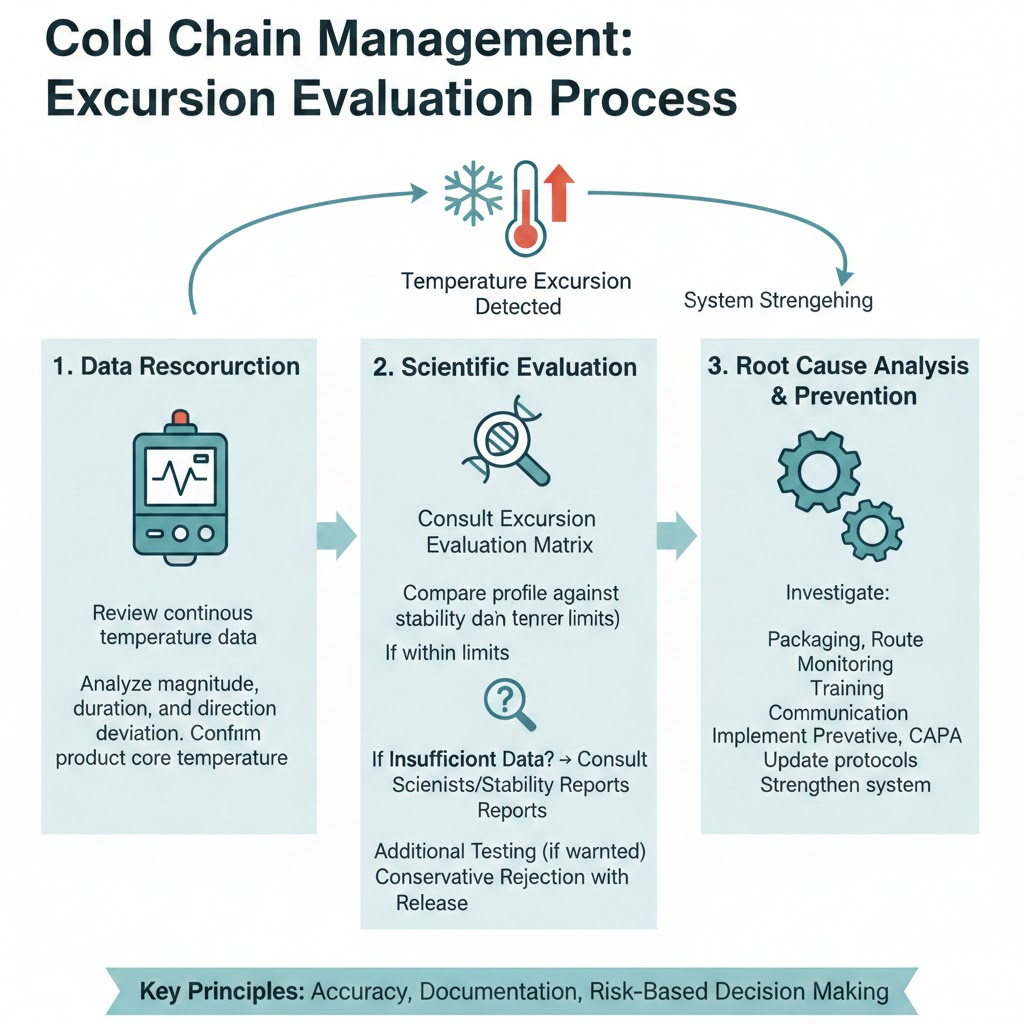
How to Scientifically Manage a Cold Chain Excursion
Effective management begins with accurate reconstruction of the event. This means reviewing continuous temperature data, understanding the duration and magnitude of deviation, and confirming whether product core temperature truly exceeded limits or whether the reading reflects external ambient conditions.
The next step involves evaluating the excursion against available stability data. Many companies maintain excursion evaluation matrices derived from real-time and accelerated stability studies. These matrices correlate specific time–temperature exposures with predicted impact. If the excursion falls within scientifically justified boundaries, the product may remain suitable for use.
Where internal data is insufficient, manufacturers may need to consult stability reports, formulation scientists, or regulatory commitments to determine appropriate action. In some cases, additional testing may be warranted. In others, conservative rejection may be the safest path.
The most important element is documentation. Regulators expect clear scientific rationale for any decision to release product following a temperature deviation. An undocumented assumption—even if scientifically correct—cannot withstand regulatory scrutiny.
Preventing Recurrence Through System Strengthening
Every cold chain excursion should prompt evaluation beyond the affected shipment. While the immediate focus is product disposition, long-term quality depends on understanding how and why the deviation occurred.
Was packaging configuration validated for the actual route duration? Were transit lanes modified without requalification? Were monitoring devices functioning correctly? Was there a delay in reviewing excursion data? Did communication between distribution partners break down?
Addressing these systemic factors reduces recurrence and strengthens overall distribution reliability. Cold chain resilience depends on proactive design, route qualification, stress testing, and continuous performance review.
Regulatory Expectations and Risk-Based Thinking
Regulatory authorities increasingly expect manufacturers to apply risk-based principles to distribution control. Good Distribution Practice frameworks emphasize maintaining product quality throughout the supply chain, not just during manufacturing. This includes validated packaging, documented excursion management procedures, and scientifically justified disposition decisions.
Importantly, regulators do not expect zero excursions. They expect controlled systems, timely investigation, and sound scientific judgment. An organization that demonstrates structured evaluation and preventive action reflects maturity in quality management.
Conclusion
Cold chain excursions are not simply logistical inconveniences. They are tests of the robustness of a company’s stability knowledge and quality systems. The ability to assess an excursion confidently depends on how well the product’s degradation behavior is understood, how accurately environmental conditions are monitored, and how disciplined the organization is in documentation and response.
What truly happens during a cold chain excursion is not always visible in a single temperature graph. It is a dynamic interaction between formulation science, exposure duration, packaging performance, and operational control. Managing these events effectively requires both scientific literacy and systemic oversight.
Ultimately, cold chain management is not about avoiding every deviation. It is about building systems strong enough to understand, evaluate, and control them when they occur. The strength of that response determines whether an excursion becomes a crisis—or simply a managed event within a well-designed quality framework.
