In pharmaceutical and life sciences manufacturing, stability studies are often viewed as a development milestone—initiated during formulation, completed to support regulatory submission, and revisited only when required. This perception is both incomplete and risky.
Stability is not a single study conducted to obtain approval; it is an ongoing scientific process that continues throughout a product’s lifecycle. From development through commercialization and market supply, stability data remains central to product quality, patient safety, and regulatory compliance.
Understanding stability as a continuous process is essential for maintaining control over product performance under real-world conditions.
The Purpose of Stability: Beyond Regulatory Approval
At its core, stability testing evaluates how a pharmaceutical product maintains its quality attributes—such as potency, purity, physical characteristics, and microbiological integrity—over time when exposed to defined environmental conditions.
While regulatory submissions require stability data to justify shelf life, storage conditions, and packaging, these data represent only a snapshot in time. They are based on limited batches, controlled conditions, and assumptions that may not fully reflect commercial reality.
Once a product enters routine manufacturing and global distribution, it is exposed to variables that were not fully present during development. Stability must therefore continue to be monitored to ensure that the product remains compliant and safe throughout its marketed life.
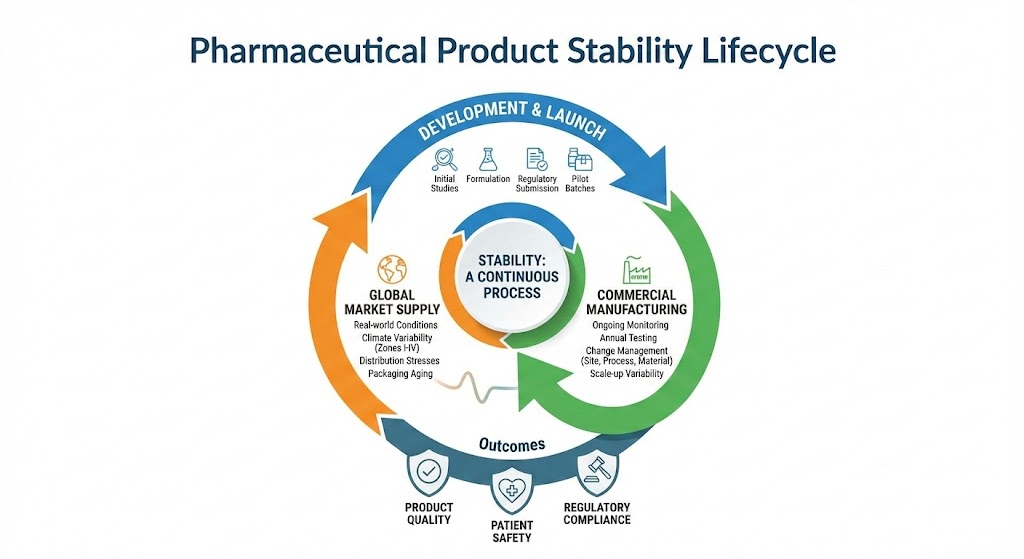
Limitations of One-Time Stability Studies
Initial stability studies are typically conducted on development or pilot-scale batches. These studies are necessary but inherently limited.
They may not capture:
- Variability introduced during large-scale manufacturing
- Changes in raw material sources or suppliers
- Equipment wear and process drift over time
- Distribution stresses across different climatic regions
- Long-term effects of packaging aging
A one-time study cannot account for cumulative risks that emerge gradually. Without ongoing evaluation, degradation trends may remain undetected until they manifest as out-of-specification results, complaints, or recalls.
Ongoing Stability Studies and Commercial Reality
Once a product is commercialized, ongoing stability studies become a regulatory and scientific necessity. These studies are designed to confirm that the shelf life and storage conditions initially approved remain valid for commercial batches.
Ongoing stability programs typically include:
- Annual or periodic stability testing of production batches
- Monitoring of long-term and accelerated conditions
- Trending of critical quality attributes over time
- Evaluation of batch-to-batch consistency
These data serve as an early warning system. Even when results remain within specification, gradual shifts in trends may indicate emerging risks that require investigation or corrective action.
Stability and Change Management
Pharmaceutical products rarely remain static after approval. Changes occur throughout the product lifecycle, including:
- Manufacturing site transfers
- Equipment upgrades
- Process optimizations
- Packaging changes
- Supplier substitutions
Each change, even when considered minor, has the potential to impact product stability. Ongoing stability data provide the scientific basis for assessing whether a change affects product quality and whether additional studies or regulatory notifications are required.
Without a continuous stability framework, organizations lack the evidence needed to justify changes confidently and compliantly.
Climate Variability and Global Distribution
Products marketed globally are exposed to a wide range of environmental conditions, particularly in hot and humid regions. Long-term exposure to high temperature and humidity can influence degradation pathways in ways that may not be fully predicted during development.
Ongoing stability testing allows manufacturers to:
- Confirm product robustness across different climatic zones
- Detect moisture- or temperature-related degradation early
- Reassess packaging performance over time
- Validate storage recommendations under real-world conditions
This is particularly critical for products distributed in ICH Climate Zone IV regions, where environmental stress is more severe and sustained.
Regulatory Expectations for Continuous Stability
Global regulatory agencies expect stability programs to extend beyond initial approval. Guidelines emphasize the importance of ongoing studies as part of Good Manufacturing Practice.
Regulators may review:
- Ongoing stability protocols and schedules
- Trending and evaluation practices
- Actions taken in response to atypical trends
- Justification for shelf life extensions or changes
Incomplete or poorly maintained stability programs can lead to regulatory observations, questions during inspections, and delays in post-approval changes.
Stability as a Risk Management Tool
When approached correctly, stability is not merely a compliance requirement but a powerful risk management tool.
Continuous stability monitoring enables manufacturers to:
- Identify degradation risks before failures occur
- Strengthen control strategies for sensitive products
- Improve packaging and storage decisions
- Protect patients from substandard or unstable products
By integrating stability data into quality systems, organizations can make informed, proactive decisions rather than reactive corrections.
Conclusion: Stability Is a Lifecycle Commitment
Stability does not end with regulatory approval or the assignment of a shelf life. It evolves alongside the product, reflecting changes in manufacturing, distribution, and market exposure.
Treating stability as a continuous process ensures that quality is maintained not only at the time of release, but throughout the entire period a product is available to patients. In an industry where safety, efficacy, and compliance are non-negotiable, ongoing stability is not optional—it is fundamental to responsible pharmaceutical manufacturing.
