India is one of the world’s largest pharmaceutical manufacturing hubs, supplying regulated and semi-regulated markets across continents. Yet many laboratories operating within the country face unique stability risks that are often underestimated—not because of poor intent, but because these risks have become normalized over time.
Stability studies are designed to prove that a product remains safe, effective, and compliant throughout its shelf life. When environmental control weakens, the science behind that proof weakens with it. In the Indian context, several systemic factors make stability assurance more complex than many labs acknowledge.
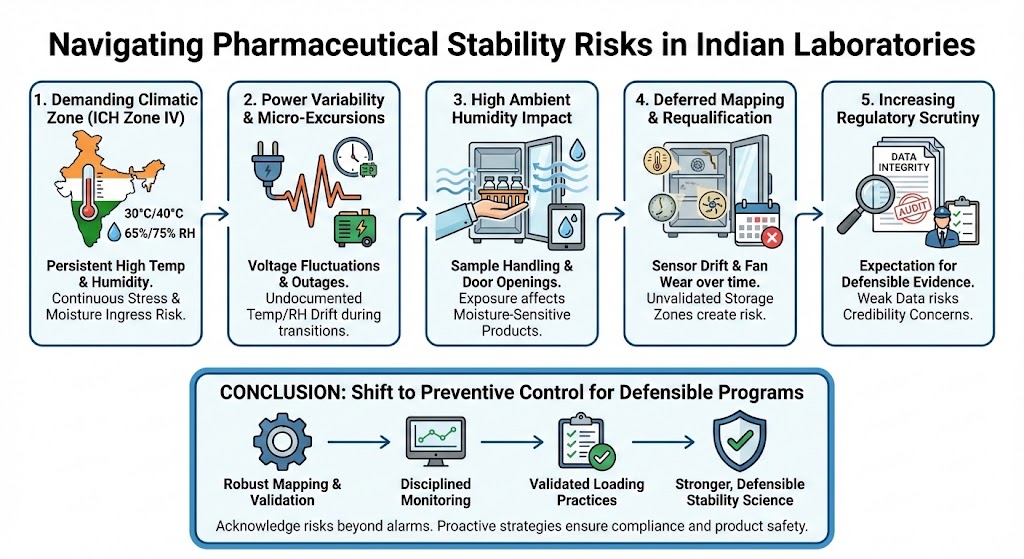
1. India Operates in the Most Demanding Climatic Zone
India falls largely within ICH Zone IV (IVa and IVb)—the most challenging stability conditions globally. Long-term and accelerated studies conducted at 30°C/65% RH and 40°C/75% RH place continuous stress on both products and equipment.
Unlike temperate regions, high humidity in India is not seasonal—it is persistent. This creates:
- Greater moisture ingress risk
- Higher condensation probability
- Increased stress on humidification and dehumidification systems
Many chambers technically meet setpoints but struggle with uniformity, recovery time, and long-term performance under sustained high humidity loads.
2. Power Variability Is a Stability Risk, Not Just an Infrastructure Issue
Voltage fluctuations, brief outages, and generator changeovers are realities in many industrial zones. While most labs plan for backup power, the transition period is often underestimated.
Short interruptions can lead to:
- Temperature and RH drift
- Slow recovery after restart
- Undocumented micro-excursions
Over time, these events may not trigger alarms but can still influence long-term stability conditions, especially in tightly controlled studies.
3. High Ambient Humidity Impacts More Than Chamber Performance
Even when a chamber performs well, ambient laboratory conditions influence outcomes:
- Sample loading and unloading
- Door openings
- Handling during pulls
- Temporary staging outside the chamber
In high-humidity environments, exposure during these moments can affect moisture-sensitive formulations and packaging—particularly tablets, capsules, and semi-solids—without leaving obvious traces in monitoring data.
4. Mapping and Requalification Are Often Deferred
In many Indian labs, mapping is performed during installation and then delayed for years unless a major failure occurs. This approach overlooks gradual changes caused by:
- Fan wear
- Sensor drift
- Shelf reconfiguration
- Increased chamber utilization
Without periodic re-mapping, laboratories may unknowingly store samples in zones that no longer represent validated conditions, weakening the scientific defensibility of stability data.
5. Regulatory Scrutiny Is Increasing, Not Decreasing
Indian manufacturers increasingly supply regulated markets, where inspectors expect:
- Strong stability rationales
- Clear chamber performance evidence
- Defensible mapping and monitoring strategies
- Data integrity across long-term studies
Questions around chamber behavior, excursions, or mapping frequency can escalate quickly from observations to data credibility concerns if supporting evidence is weak.
Conclusion
Indian laboratories operate under some of the most demanding environmental conditions in the global pharmaceutical ecosystem. The risks they face are not due to lack of expertise or effort—but due to climate, infrastructure variability, high utilization, and evolving regulatory expectations.
Recognizing these risks early allows laboratories to shift from reactive fixes to preventive control—through robust mapping strategies, disciplined monitoring, validated loading practices, and realistic environmental planning.
In stability science, the absence of alarms does not always mean the absence of risk.
For Indian labs, acknowledging that reality is the first step toward stronger, more defensible stability programs.
